Abstract
The Museo Egizio in Turin holds a unique collection of 116 mummified or skeletal human bodies or body parts. This collection is currently under study by the “Mummy Conservation Project”, a collaborative venture of the Museo Egizio, the Institute for Mummy Studies of Eurac Research, the Soprintendenza Archeologia del Piemonte, and the Horus Group, whose aim is to improve mummy conservation techniques. In the context of this project, an assessment of the state of preservation of the mummified human remains was carried out by monitoring basic physical parameters (temperature, relative humidity and water activity) and by performing a fungal survey. The latter revealed the presence on the mummified material of fungal spores and mycelia that could possibly pose a biodegradative threat. However, all the current physical parameters show that the mummies are stored under optimal environmental conditions, which will suppress any microbial up-growth.
1. Introduction
The first set of Egyptian mummies arrived in Turin following the scientific/commercial expedition undertaken by the traveler and naturalist, Vitaliano
Finally, thanks to Ernesto Schiaparelli, director of the Museo Egizio between 1894 and 1928, the number of mummies in the collection was further increased. A buying trip to Egypt, done by Schiaparelli in 1900/1901, led to the purchase of more than 1,500 items, including two naturally mummified adult bodies and the mummy of a small child dating to about 3500 BCE. But it is especially since the foundation of the Italian Archaeological Mission to Egypt in 1903 that a large number of human remains was discovered at various sites along the Nile, during many excavation seasons directed by Egyptologists Ernesto Schiaparelli and, later on, Giulio Farina, who were assisted, since 1914, by anthropologist
Given the increasing attention among researchers to the mummified human remains held in the Museo Egizio in Turin, in 2016 the “Mummy Conservation Project” was set up, with the aim of incorporating the data and knowledge acquired so far on these human remains in order to develop a protocol for proper museum conservation and display.
The goals of the “Mummy Conservation Project” illustrated in this paper were the following:
-
Taking thermo-hygrometric measurements in the galleries, the inside of display cases, in the mummy storage room, and in the room where the
research operations were carried out. - Measuring the water activity (aw) of the mummies.
- Fungal spore sampling of the air in the galleries, inside a display case, in the mummy storage room, and in the room where the research operations were carried out.
- Fungal survey of the mummified material.
2. Research aim
In view of the Museo Egizio and Eurac Research’s common interest in developing know-how to help preserve all the Museo Egizio’s mummies – both those on display and those in storage – special emphasis was placed on testing water activity (aw), a parameter that affects the rate of biochemical decay in organic tissues. This important parameter is significantly affected by temperature and relative humidity in the conservation environment. In parallel to the assessment of this physical parameter, a first microbial survey was performed.
To identify the presence of possible superficial growing fungi, samples of both textiles and skin were taken using non-invasive contact plates.
To obtain further knowledge of the environmental conditions the mummies are presently experiencing, the researchers also took samples of spores present in the air inside the storage room where the mummies were kept, in a display case in the permanent galleries, and in the room where the research operations were carried out.
The aim of this work is to expand on research on the conservation organic objects of historical and artistic interest by assessing both biological and physical-chemical conservation requirements.
3. Material and methods
3.1 Thermo-hygrometric measurement of the exhibition areas, inside a display case, in the mummy storage room, and in the room where the research operations were carried out
Thanks to historical data relative to the thermo-hygrometric conditions of the museum’s exhibition spaces and the storage space where the mummies were kept (prior to the inauguration of the new galleries in 2015), it was possible to identify the seasonal
With the exception of the room where the Tomb of Kha and Merit was display – where a wall-mounted air conditioner and a portable humidifier were installed – all the rest of the museum and storage lacked an air conditioning system. Climate monitoring took place in “sample” mode over different periods and in different areas of the museum with the aid of data loggers (HumiStick). The recorded temperature ranged between 21 °C and 30 °C, and the recorded relative humidity between 30 % and 66 %.
In 2015, a new air conditioning system was installed, serving all the exhibition halls, the Foundation’s offices, and the storage spaces. It is powered by an underground thermo-cooling plant using geothermal energy.
To date, the museum’s different exhibition areas have depended on auxiliary machines whose temperature and relative humidity values can be individually and independently set.
3.2 Measuring water activity (aw) of the mummies
As regards this specific phase of the project, a total of 106 organic samples were analysed, both of a vegetal nature (bandage linen) and of human origin (fragments of bone, skin, muscles, connective tissue, hair)(Fig. 1).
A phase of a non-invasive water activity (aw) test carried out on the mummy of Kha (Suppl. 8431). Picture by Eurac Research, Institute for Mummy Studies.
To measure the water activity (aw) value, the Rotronic instrument, model HygroPalm HP-23-AW-A, was used. Prior to measuring the samples, the HC2-AW and the relative Rotronic HC2-SH measurement probe were calibrated as needed.
Immediately after sampling the mummified organic tissues, the water activity of the samples was examined. The samples were then placed inside special sample holders, previously sterilised. The Rotronic HW4 software version 3.8.0 was used to validate the measurements obtained. Once the PC was connected to the analyser, it was possible, through this application, to set the necessary operating settings in order to obtain the measurement (Fig. 2).
Simultaneously with the water activity (aw) acquisition phase, the temperature and relative humidity values of the environment were obtained. The time required to acquire each sample is approximately 5 minutes. At the end of each acquisition, a validation certificate was produced in form of a PDF file.
Instrumental configuration for assessing water activity (aw). Picture by Marco Samadelli/Eurac Research, Institute for Mummy Studies.
3.3 Sampling and cultivation of fungi
Non-invasive sampling using contact agar plates (DG18 and 2 % MEA, Merck) was performed as previously
Direct microbiological sampling on the surface of mummy Cat. 2245. Picture by Marco Samadelli/Eurac Research, Institute for Mummy Studies.
Sampling of spores present in the air inside display cases (Suppl. 19691). Picture by Marco Samadelli/Eurac Research, Institute for Mummy Studies.
3.3.1 DNA extraction, PCR amplification and sequence analysis
Pure cultures were identified based on the sequencing of internal transcribed spacer region (ITSI-5.8S-ITSII), a sequence that can be used as a phylogenetic marker. DNA was extracted from the purified cultures using fresh mycelia, as described by Michaelsen et al.7 For the analysis of fungal ITS sequences, we PCR amplified this genomic region using specific ITS primers,8 using the conditions described by Pinar et al.9 To identify the fungi, we compared the obtained sequence material to fungi ITS sequences available in the online public database NCBI, using the BLAST search program.10
4. Results and discussion
4.1 Observations on the results of water activity (aw) analysis
Water activity analysis is a widely-used technique in food storage control.11 Its application in the field of cultural heritage conservation, on mummified biological samples, is innovative and pioneering.
The importance of relative humidity (RH) to conservation is immense,12 and closely linked to the concept of water activity (aw), the parameter proportional to the escaping tendency of the water molecules present in the tissues constituting the mummy. Water activity (aw) is calculated as the ratio of the partial vapour pressure of water in tissues (Pw) to that of pure water (Pw0) at the sample’s surface temperature.13
After a certain time, any substance placed in a sealed
From the chemical point of view, tissues constituting mummies can be considered as concentrated “solutions”, and measurement and limitation of water activity (aw) are essential to the conservation process because they influence the life of microorganisms and enzyme activity. The greater the water activity (aw) value, the faster the decay of organic tissue, as synthetically represented in the chart (Fig. 5).15
Positioning of the results of water activity (aw) testing on the mummies in relation to the decay speed of organic tissues as a function of water activity (aw) [Labuza, T.P., 1975].![Positioning of the results of water activity (aw) testing on the mummies in relation to the decay speed of organic tissues as a function of water activity (aw) [Labuza, T.P., 1975].](https://rivista.museoegizio.it/wp-content/uploads/2019/11/fig-5-site.jpg)
Relative humidity values higher than 65 %, combined with temperature values higher than 20 °C, promote the development of moulds and accelerate the metabolism of many harmful insects. It is scientifically demonstrated that, with water activity (aw) lower than 0.3, it is possible to inhibit the majority of the causes of biochemical decay in organic tissue.16
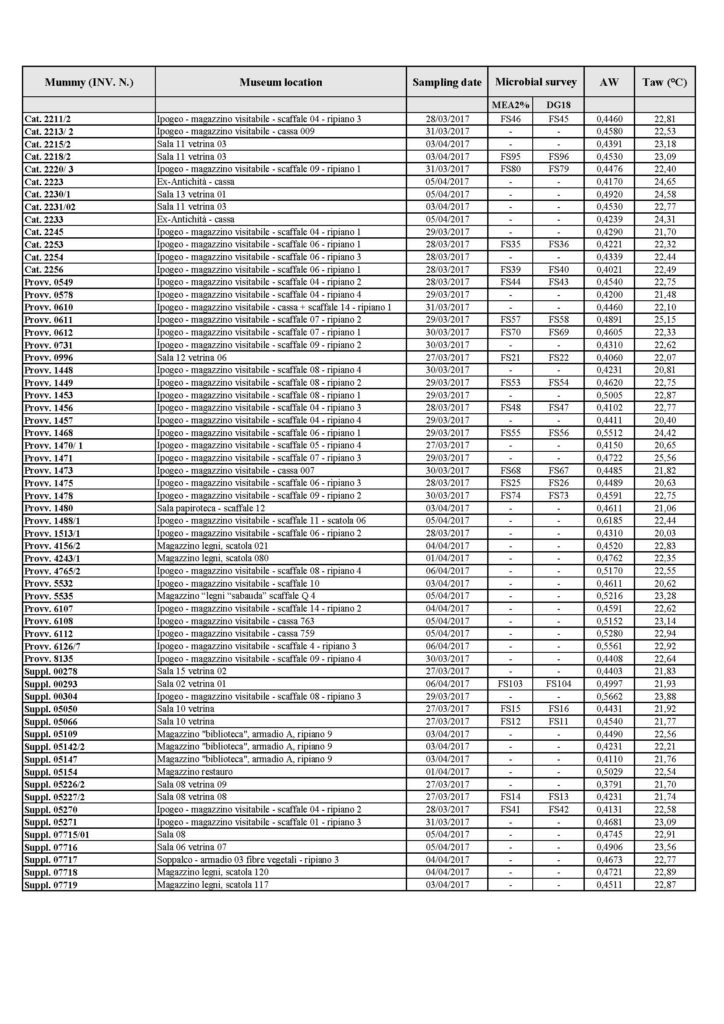
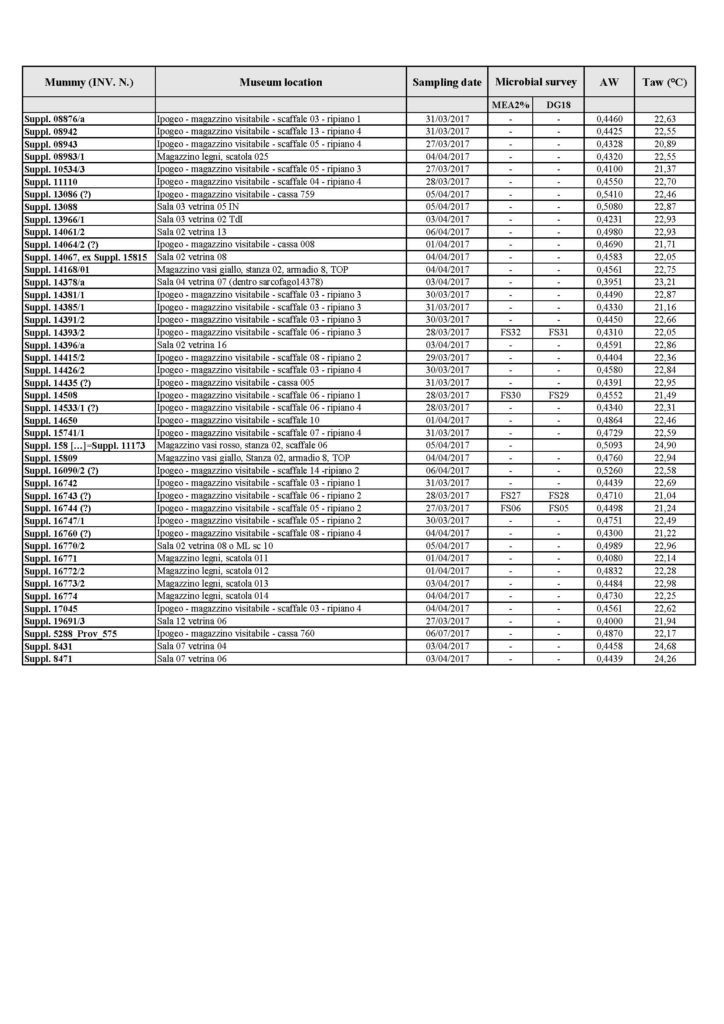
A total of 106 mummified samples were subjected to water activity (aw) tests. The results of the analysis are illustrated in Tables 1A and 1B (Tab. 1A and Tab. 1B).
The results of the water activity (aw) value for the mummies, correlated with the acquisition temperature value Taw (°C) of the room where the research operations were carried out.
Results of the water activity (aw) value for the mummies, correlated with the acquisition temperature value Taw (°C) of the room where the research operations were carried out.
The results of our water activity (aw) examination shows that most of the analysed mummies are in the range between 0.4 and 0.5 (aw) (Fig. 6). No differences were noted, with respect to this type of analysis, between one mummy and the other, even when kept in different environments. This uniformity of results is not surprising, because the same homogeneity was observed in the climatic parameters of the museum’s exhibition environments and display cases, and of the mummy storage room.
Relationship between the number of mummies and the results of the water activity (aw) analysis.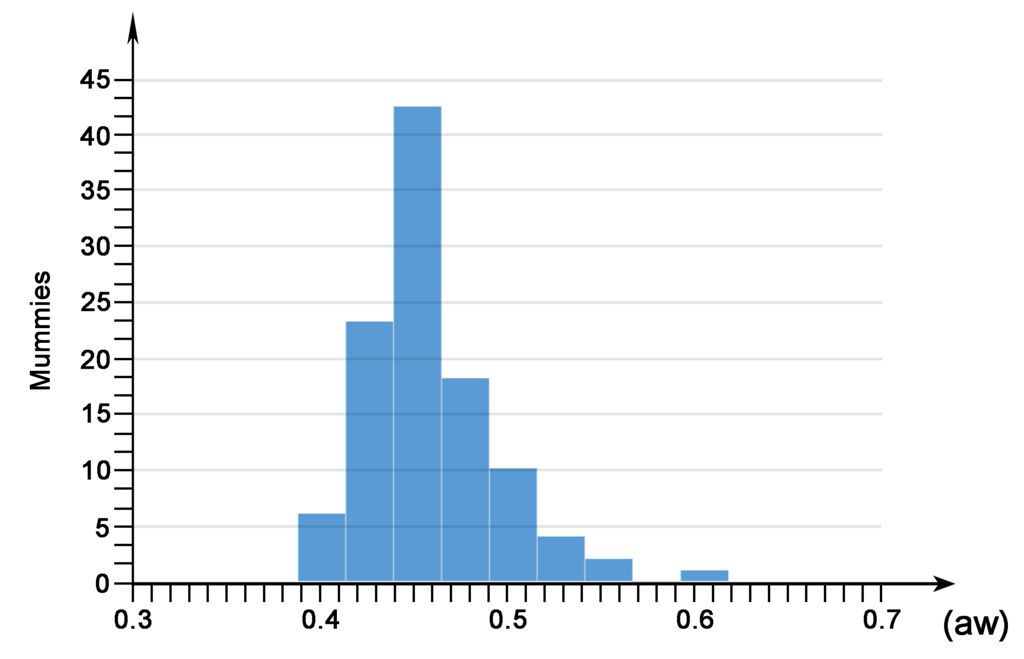
In conclusion, observing the results of the water activity (aw) analysis on the mummified samples, reported in tables 1A and 1B (Tab. 1A and Tab. 1B), and comparing them with recent theoretical and experimental studies relative to the conservation of organic mummified finds,17 it can be stated that:
- the observed water activity levels are consistent with those expectable in the environmental climatic conditions provided by the museum,
- the analysed mummies are preserved within the safety parameters provided for proper conservation, as they all remain within the ideal range between 0.25 – 0.65 (aw), as shown in Fig. 5.18
4.2 Microbiological analysis
Through the fungal survey applied in this study, we were able to detect and identify different fungal genera and species on skin and textile samples taken from mummies from both the storage rooms and the display cases (Fig. 7). Interestingly, the number of
Fungi detected on textiles and soft tissues of mummies from the storage room (Ipogeo, magazzino visitabile) and from display cases. The pictures on the upper right show a contact plate sampling on a grass mat and the fungal growth on the agar plate after incubation. The fungi marked with an asterisk have been detected both on the contact plates and in the air sampling.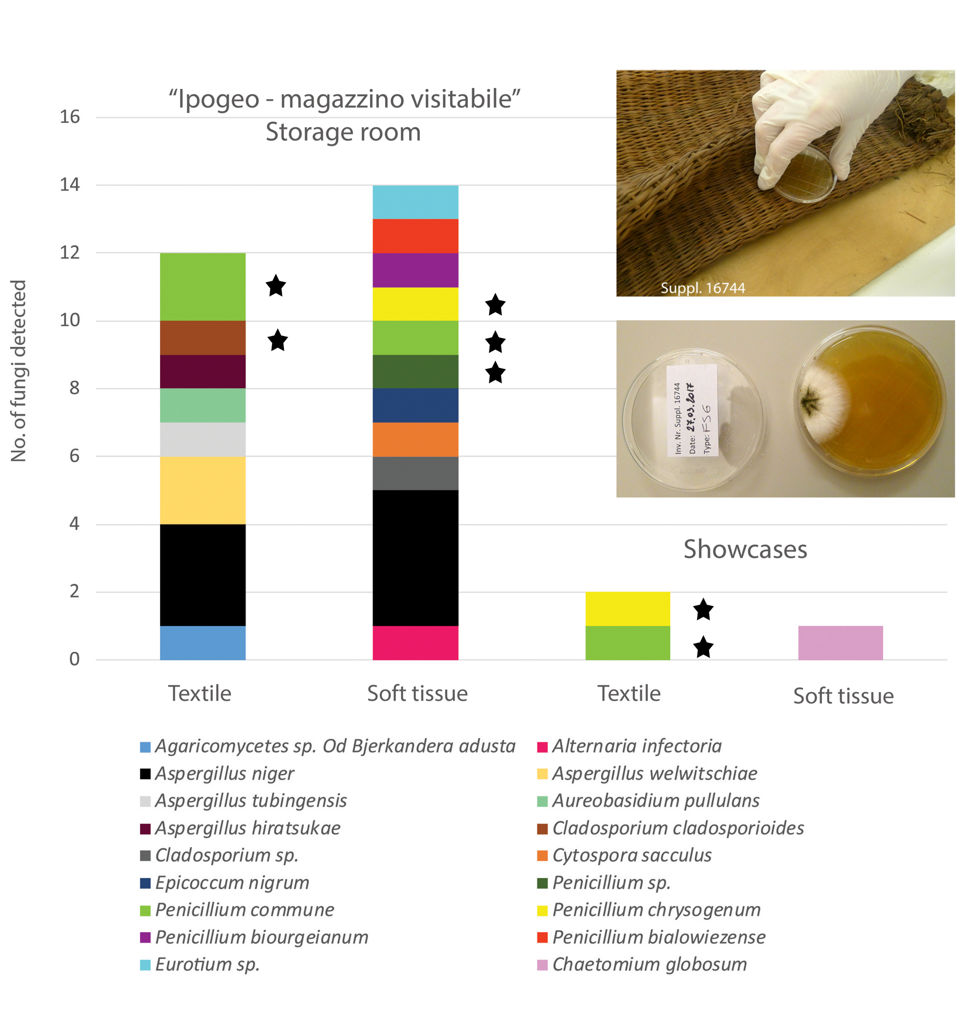
5. Final considerations
In sum, our results indicate a still present biodegradational potential in the form of tissue-specific fungal communities (spores and mycelia), which could become a biodegradative risk as soon as the environmental conditions change, for example, at water activity levels above 0.65 (aw), which promote fungal growth.23 It is therefore of the utmost importance to keep the water activity levels of these unique cultural assets under constant control.
6. Acknowledgements
The authors wish to thank their partners for their valuable collaboration, without which it would not have been possible to complete this study project: the director of the Museo Egizio Christian Greco, Sara Aicardi, Susanne Töpfer, Valentina Turina,
Bibliography
Altschul, S.F., T.L. Madden, A.A. Schaffer, J. Zhang, Z. Zhang, W. Miller, and D.J. Lipman, “Gapped BLAST and PSI-BLAST: A New Generation of Protein Database Search Programs”, Nucleic Acids Research 25/17 (1997), pp. 3389–402.
Bensch, K., J.Z. Groenewald, J. Dijksterhuis, M. Starink-Willemse, B. Andersen, B.A. Summerell, H.D. Shin, F.M. Dugan, H.J. Schroers, U. Braun, and P.W. Crous, “Species and Ecological Diversity Within the Cladosporium Cladosporioides Complex (Davidiellaceae, Capnodiales)”, Studies in Mycology 67 (2010), pp. 1–94.
Boano, R., R. Grilletto, A.M. Donadoni Roveri, E. Rabino Massa, and E. Fulcheri, “Le mummie del Museo Egizio di Torino: indagini diagnostiche preliminari sullo stato di conservazione”, in F. Barbagli (ed.), Preparazione, conservazione e restauro dei reperti naturalistici: metodologie ed esperienze. Atti dei Seminari ANMS di Pavia, Museologia Scientifica Memorie 3 (2008), pp. 107–11.
Boano, R., G. Mangiapane, M. Girotti, and E. Rabino
Boano, R., F. Cesarani, G. Gandini, R. Grilletto, E. Fiore Marocchetti, and M. C. Martina, “Mummie e mummificazione”, in Museo Egizio, Modena 2015, pp. 244–51.
Cheftel, J.-C., H. Cheftel, P. Besançon, and L. Didero (eds.), Biochimica e tecnologia degli alimenti, Bologna 19992, pp. 480–84.
Delorenzi, E., R. Grilletto, Le mummie del Museo Egizio di Torino. N. 13001–13026. Indagine antropo-radiologica (Catalogo generale del Museo Egizio di Torino, serie seconda – collezioni 6), Milano 1989, pp. 17–46.
Duarte, E.A.A., C.L. Damasceno, T.A.S. De Oliveira, L.D.O. Barbosa, F.M. Martins, J.R. De Queiroz Silva, T.E.F. De Lima, R.M. Da Silva, R.B. Kato, D.E. Bortolini, V. Azevedo, A. Gòes-Neto, and A.C.F. Soares, “Putting the Mess in Order: Aspergillus Welwitschiae (and not A. Niger) is the Etiological Agent of Sisal Bole Rot Disease in Brazil”, Frontiers in Microbiology 9 (2018).
Dubois, D., M. Pihet, C.L. Clech, A. Croue, H. Beguin, J.P. Bouchara, and D. Chabasse, “Cutaneous Phaeohyphomycosis due to Alternaria Infectoria”, Mycopathologia 160 (2005), pp. 117–23.
EN 15803, Conservation of Cultural Property – Test Methods – Determination of Water Vapour Permeability, European Committee for Standardization (CEN), 2009.
Fan, X., K.D. Hyde, M. Liu, Y. Liang, and C. Tian, “Cytospora Species Associated with Walnut Canker Disease in China, with Description of a New Species C. Gigalocus”, Fungal Biology 119/5 (2015), pp. 310–19.
Fiore Marochetti, E., “Le mummie delle collezioni Donati e Drovetti al Museo delle Antichità Egizie di Torino”, in R. Boano and E. Rabino Massa (eds.), Mummie Egizie in Piemonte. Storia ed attualità in ambito egittologico ed antropologico, Torino 2012, pp. 5-6.
FprEN 16242, Conservation of Cultural Property – Procedures and Instruments for Measuring Humidity in the Air and Moisture Exchanges Between Air and Cultural Property (under approval), European Committee for Standardization (CEN), 2012.
Labuza, T.P., “Sorption Phenomena in Foods: Theoretical and Practical Aspects”, in: Cho-Kyun Rha (ed.), Theory, Determination and Control of Physical Properties of Food Materials, Food Material Science, I (1975), pp. 197–219.
Marro, G., “Scavi Italiani in Egitto e loro scopo antropologico”, XV Congrès International d’Anthropologie & d’Archéologie Préhistoríque, IV Session de l’Institut International d’Anthropologie, Portugal, 21-30 septembre 1930, Paris 1931, pp. 1–8.
Martín, A., M.A. Asensio, M.E. Bermúdez, M.G. Córdoba, E. Aranda, and J.J. Córdoba, “Proteolytic Activity of Penicillium Chrysogenum and Debaryomyces Hansenii During Controlled Ripening of Pork Loins”, Meat Science 62 (2002),
Moiso, B., La storia del Museo Egizio, Torino and Modena 2016, pp. 40–55.
Michaelsen, A., F. Pinzari, K. Ripka, W. Lubitz, and G. Piñar, “Application of Molecular Techniques for Identification of Fungal Communities Colonising Paper Material”, International Biodeterioration & Biodegradation 58/3 (2006), pp. 133–41.
Perrone, G., G. Mulè, A. Susca, P. Battilani, A. Pietri, and A. Logrieco, “Ochratoxin A Production and Amplified Fragment Length Polymorphism Analysis of Aspergillus Carbonarius, Aspergillus Tubingensis, and Aspergillus Niger Strains Isolated from Grapes in Italy”, Applied Environmental Microbiology 72/1 (2006), pp. 680–85.
Piñar, G., D. Piombino-Mascali, F. Maixner, A. Zink, and K. Sterflinger, “Microbial Survey of the Mummies from the Capuchin Catacombs of Palermo, Italy: Biodeterioration Risk and Contamination of the Indoor Air”, FEMS Microbiology Ecology 86/2 (2013), pp. 341–56.
Piñar, G., L. Krakova, D. Pangallo, D. Piombino-Mascali, F. Maixner, A. Zink, and K. Sterflinger, “Halophilic Bacteria Are Colonizing the Exhibition Areas of the Capuchin Catacombs in Palermo, Italy”, Extremophiles 18/4 (2014), pp. 677–91.
Samadelli, M., G. Roselli, V.C. Fernicola, L. Moroder, and A.R. Zink, “Theoretical Aspects of Physical-Chemical Parameters for the Correct Conservation of Mummies on Display in Museums and Preserved in Storage Rooms”, Journal of Cultural Heritage 14/6 (2013), pp. 480–84.
Scattolin Morecroft, A., “The Vitaliano Donati Collection at the Turin Egyptian Museum”, JEA 92 (2006), pp. 278-82.
Shin, M., Oxygen-free Museum Cases (series II – Research in Conservation), Getty Conservation Institute, 1998.
Wang, Y., R. Vazquez-Duhalt, and M.A. Pickard, “Manganese-Lignin Peroxidase Hybrid from Bjerkandera Adusta Oxidizes Polycyclic Aromatic Hydrocarbons More Actively in the Absence of Manganese”, Canadian Journal of Microbiology 49/11 (2003), pp. 675–82.
White, T., T. Bruns, S. Lee, and J. Taylor, “Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics”, in: M.A. Innis, D.H. Gelfand, J.J. Shinsky, and T.J. White (eds.), PCR Protocols: A Guide to Methods and Applications, San Diego 1990, pp. 315–22.
Notes
- Scattolin Morecroft, in <i>JEA</i> 92 (2006), pp. 278, 281.↑
- Moiso, <i>La storia del Museo Egizio</i> (2016).↑
- For an overview on the mummies from Donati’s and Drovetti’s collection, see Fiore Marochetti in Boano and Rabino Massa (eds.), <i>Mummie Egizie in Piemonte</i> (2012), pp. 5-6; Boano et al., <i>Mummie e mummificazione</i> (2015); Delorenzi, Grilletto, <i>Le mummie del Museo Egizio di Torino</i> (1989).↑
- Boano et al., <i>Le mummie del Museo Egizio di Torino</i> (2008); Marro, <i>Scavi Italiani in Egitto</i> (1930).↑
- Boano et al. in Boano and Rabino Massa (eds.), <i>Mummie Egizie in Piemonte</i> (2012), p. 21.↑
- Piñar et al., <i>FEMS Microbiology Ecology</i> 86/2 (2013); Piñar et al., <i>Extremophiles</i> 18/4 (2014).↑
- Michaelsen et al., <i>International Biodeterioration & Biodegradation</i> 58/3 (2006).↑
- White et al., in Innis et al. (eds.), <i>PCR Protocols: A Guide to Methods and Applications</i> (1990).↑
- Piñar et al., <i>FEMS Microbiology Ecology</i> 86/2 (2013).↑
- Altschul et al., <i>Nucleic Acids Research</i> 25/17 (1997).↑
- Cheftel et al. (eds.), <i>Biochimica e tecnologia degli alimenti</i> (1999).↑
- FprEN 16242.↑
- EN 15803.↑
- EN 15803.↑
- Labuza, in Cho-Kyun Rha (ed.), <i>Theory, Determination and Control</i>, 1975.↑
- Labuza, in Cho-Kyun Rha (ed.), <i>Theory, Determination and Control</i>, 1975; Shin, <i>Oxygen-Free Museum Cases</i>, 1998; Samadelli et al., <i>Journal of Cultural Heritage</i> 14/6 (2013).↑
- Samadelli et al., <i>Journal of Cultural Heritage</i> 14/6 (2013).↑
- Labuza, in Cho-Kyun Rha (ed.), <i>Theory, Determination and Control</i>, 1975.↑
- Bensch et al., <i>Studies in Mycology</i> 67 (2010); Fan et al., <i>Fungal Biology</i> 119/5 (2015); Perrone et al., <i>Applied Environmental Microbiology</i> 72/1 (2006); Wang et al., <i>Canadian Journal of Microbiology</i> 49/11 (2003).↑
- Duarte et al., <i>Frontiers in Microbiology</i> 9 (2018).↑
- Dubois et al., <i>Mycopathologia</i> 160 (2005).↑
- Martín et al., <i>Meat Science</i> 62 (2002).↑
- Labuza, in Cho-Kyun Rha (ed.), <i>Theory, Determination and Control</i> (1975).↑